Dmitri Ivanovich Mendeleev (1834 – 1907) was a Russian chemist most famous for his contributions to the Periodic Table. He was the first to publish a periodic table similar to the one we use today and is credited for discovering the Periodic law. More importantly, he put the elements in their right places in his periodic table correctly pointing out that their atomic weights had been measured incorrectly; and brilliantly predicted the existence and properties of yet undiscovered elements. It was due to Mendeleev’s genius that the Periodic Table was popularized in the scientific community and hence he is regarded as the Father of the Periodic Table. Mendeleev’s other accomplishments include important contributions in determining the nature of solutions; defining the critical temperature of gases; work in transforming the petroleum industry in Russia; and introducing the metric system in Russia. Know more about the work of Dmitri Mendeleev through his 10 major contributions.
#1 HE WROTE A RENOWNED TEXTBOOK ON CHEMISTRY
Dmitri Mendeleev was worried that chemistry in Russia was trailing behind the science in Europe. In 1861, to address the issue, he published a textbook named Organic Chemistry which won him the prestigious Domidov Prize and put him at the forefront of Russian chemical education. Mendeleev was one of the founders, in 1869, of the Russian Chemical Society. His renowned work Osnovy khimii (The Principles of Chemistry, 1868–71) was published in two volumes. The Principles of Chemistry became the definitive textbook on the subject at the time, ran through many editions and was widely translated. Mendeleev later rated it as one of his four major contributions to science; the others being the periodic table, the elasticity of gases, and understanding of solutions as associations.

#2 HE WAS FIRST TO PUBLISH A PERIODIC TABLE AKIN TO THE MODERN ONE
Though several earlier attempts had been made to classify the chemical elements, the first person to arrange the elements in order of their relative atomic masses (then called atomic weights) and notice their periodicity was French geologist A.E.Béguyer de Chancourtois, who did so in 1862. However, as he used geological terms and as his chart included ions and compounds, his publication was ignored by chemists. Many other chemists made significant progress in the formulation of a periodic table, most notably John Newlands. However, it was Dmitri Mendeleyev who first published a periodic table similar to the modern one we use today, in 1869. German chemist Julius Lothar Meyer independently arrived at a periodic table similar to Mendeleev but he published it a year later.
#3 HE INDEPENDENTLY DISCOVERED THE PERIODIC LAW
On 6 March 1869, Dmitri Mendeleev presented the first periodic table to the Russian Chemical Society. In his presentation, which was entitled The Dependence between the Properties of the Atomic Weights of the Elements, he described chemical elements according to both atomic weight and valency. He stated several important points during the presentation including the Periodic law, which states that when elements are ordered according to their atomic weights, certain properties of elements repeat periodically. Mendeleev’s periodic table organized all known elements according to their atomic weights and was a visual representation of the periodic law. Though other scientists, like Newlands, also noted periodicity of elements, the credit of the discovery is given to Mendeleev and Meyer. Mendeleev arrived at the law independently from investigations of other scientists.
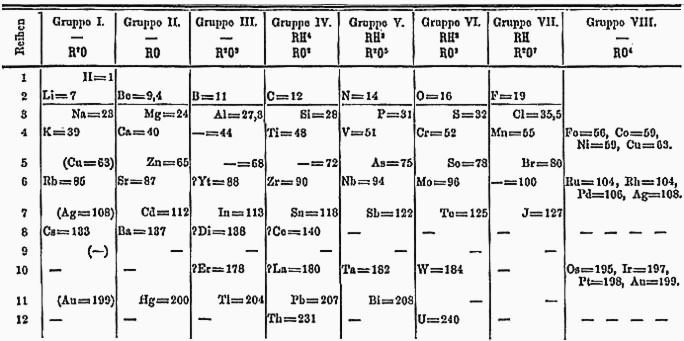
#4 HE CORRECTLY PREDICTED THAT ATOMIC WEIGHTS OF SOME ELEMENTS WERE WRONG
Unlike other chemists, Mendeleev put the elements of the periodic table in their correct places. At the time, atomic weights were determined by multiplying equivalent weight with valency. Sometimes these were incorrect due to wrong valency assigned to an element. Like beryllium was given a valency of 3 due to which its atomic weight came out to be 13.8. However Mendeleev said that the valency was 2 to fit it into the space between Li and B. Similarly, Mendeleev proposed that atomic weights of some elements had been measured incorrectly and his predictions soon turned out to be true! He was wrong at this at times because, though he had placed the elements in his table at their correct positions, it is atomic number and not atomic weight, that governs an element’s position in the Periodic Table; but in most cases the two result in the same order.
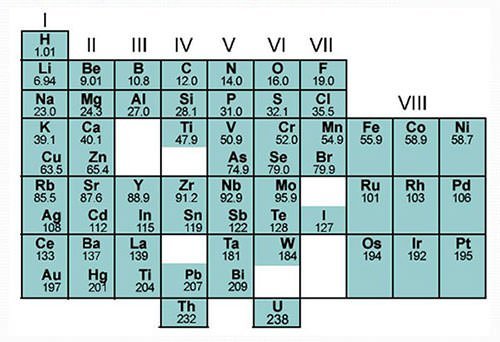
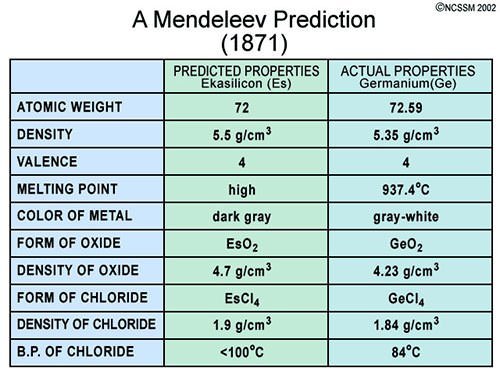
#5 HE CORRECTLY PREDICTED THE EXISTENCE OF UNDISCOVERED ELEMENTS
The most spectacular accomplishments of Mendeleev was that he not only left gaps in his periodic table for elements which were not yet discovered but more importantly predicted the properties of some of these elements and their compounds. Three of these elements were discovered within 15 years while Mendeleev was alive. He had named them eka-aluminium, eka-boron and eka-silicon after the elements which they followed (eka = 1 in Sanskrit). In 1875, French chemist Paul-Émile Lecoq de Boisbaudran discovered gallium, predicted by Mendeleev as eka-aluminium; in 1879 Swede Lars Nilson identified scandium, Mendeleev’s eka-boron; and in 1886, German Clemens Winkler discovered germanium, Mendeleev’s eka-silicon. After Mendeleev’s death, his dvi-manganese (dvi = 2 in Sanskrit) and eka-manganese, technetium and rhenium, were discovered in 1926 and 1937 respectively.
#6 MENDELEEV IS CONSIDERED THE FATHER OF THE PERIODIC TABLE
Though many other scientists made important contributions in the development of the Periodic Table, Dmitri Mendeleev was the first chemist to use the trends in his periodic table to correctly predict the properties of missing elements, such as gallium and germanium; and to ignore the order suggested by the atomic weights of the time, to better classify the elements into chemical families. Also, as his predictions started to come true, more and more people took notice of his work helping in establishing the importance of the Periodic Table. Due to all these achievements, Dmitri Mendeleev is referred to as the Father of the Periodic Table.

#7 HE MADE IMPORTANT CONTRIBUTIONS IN DETERMINING THE NATURE OF SOLUTIONS
Dmitri Mendeleev devoted much time to the study of such “indefinite” compounds as solutions. He considered solutions as liquid systems in a state of dissociation. According to him, these systems consist of molecules of the solvent and solute and of products of their interaction. His views on the nature of solutions and vast experimental data concerning the same were presented in his 1887 monograph Study of Aqueous Solutions From Their Specific Gravity. In the monograph, he anticipates the theory of hydration of ions. His ideas regarding the chemical interaction among the components of a solution contributed significantly to the development of the modern theory of solutions.

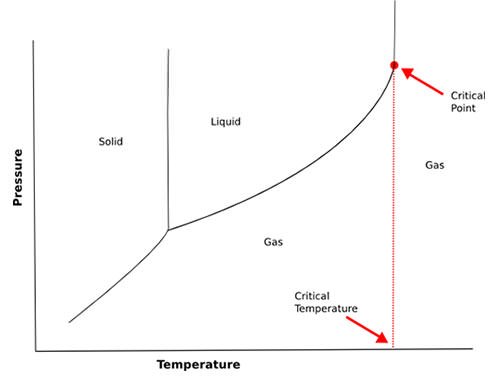
#8 HE DEFINED CRITICAL TEMPERATURE OF GASES
In the field of physical chemistry, Dmitri Mendeleev investigated the expansion of liquids with heat. He devised a formula for the expansion of liquids with heat which was similar to Gay-Lussac’s law of the uniformity of the expansion of gases. The critical temperature of a gas is the temperature above which it cannot be liquefied by any amount of pressure. It was first discovered by French physicist Charles Cagniard de la Tour. Mendeleev worked in the area and anticipated Irish scientist Thomas Andrews’ conception of the critical temperature of gases by defining the critical temperature of a substance as the temperature at which cohesion and heat of vaporization become equal to zero and the liquid changes to vapor, irrespective of the pressure and volume.
#9 HE MADE IMPORTANT CONTRIBUTIONS TO THE PETROLEUM INDUSTRY IN RUSSIA
A prominent feature of Mendeleev’s scientific career was that it aligned with the economic development of Russia. Mendeleev was particularly interested in petroleum, coal, metallurgical and chemical industries. He investigated the composition of petroleum, put forward the hypothesis that it was formed deep within the earth and predicted that it will become a key component of the world economy. Mendeleev helped in the formation of the first oil refinery in Russia and was also the first to suggest the idea of using pipelines for transportation of fuel in 1863.

#10 HE IS CREDITED WITH INTRODUCING THE METRIC SYSTEM IN RUSSIA
Mendeleev conducted studies in the field of metrology, the scientific study of measurement. He developed a precise theory of weights; designed an excellent balance arm and arresting device; and proposed highly exact methods of weighing. Mendeleev is given credit for the introduction of the metric system in Russia. At his insistence the system was made optional in 1899, but it was only in 1918, after his death, that it was made mandatory. The D.I. Mendeleev All-Russian Institute for Metrology (VNIIM), one of the largest world centers of scientific and practical metrology, is named after Mendeleev. On the behest of the Russian navy, Mendeleev also invented a smokeless powder named pyrocollodion, to replace gunpowder. However it was not used due to its cost of production.

Thank you so much! This really helped me out a lot. Thank you again for your cooperation!
You’re welcome and happy to help you.